Using AI to develop hydrogen fuel cell catalysts more efficiently and economically
October 19, 2023
Source: Nanowerk News
| Oct 18, 2023 | |
Using AI to develop hydrogen fuel cell catalysts more efficiently and economically | |
| (Nanowerk News) Proton exchange membrane hydrogen fuel cells (PEMFCs) used in hydrogen vehicles use expensive platinum catalysts to facilitate the oxygen reduction reaction at the anode. There are a vast number of elemental combinations and compositions that need to be explored to develop more efficient and cost-effective catalyst materials than platinum, and researchers are still doing a lot of trial and error in the lab. | |
Key Takeaways | |
| |
| Graphical abstract of machine learning-driven hydrogen fuel cell catalyst design. (Image: KIST) | |
The Research | |
| The Korea Institute of Science and Technology (KIST) announced that Dr. Donghun Kim and Dr. Sang Soo Han of the Computational Science Research Center, Dr. Jong Min Kim of the Materials Architecturing Research Center, and Prof. Hyuck Mo Lee of the Department of Materials Science and Engineering at the Korea Advanced Institute of Science and Technology (KAIST) have presented a new artificial intelligence-based catalyst screening methodology and succeeded in developing a new catalytic material based on a ternary element-based alloy (Cu-Au-Pt) that is cheaper and performs more than twice as well as pure platinum catalysts. | |
| The research has been published in Applied Catalysis B: Environmental ("Machine learning filters out efficient electrocatalysts in the massive ternary alloy space for fuel cells"). | |
| The team developed Slab Graph Convolutional Neural Network (SGCNN) artificial intelligence model to accurately predict the binding energy of adsorbates on the catalyst surface. This is not the first application of AI to materials discovery. The SGCNN model was developed by evolving the CGCNN model, which is specialized in predicting bulk properties of solid materials, to predict surface properties of catalytic materials. | |
| However, there is a big difference between predicting bulk properties and surface properties. When you can quickly and accurately predict the surface properties of a catalyst, you can more efficiently screen for catalysts that meet the triple bottom line of material stability, performance, and cost. In fact, when developing fuel cell anode reaction catalysts using this methodology, we were able to explore the potential of nearly 3,200 ternary candidate materials in just one day, a scale that would have taken years using the density functional theory (DFT) adsorption energy simulation calculations traditionally used to predict catalyst properties. | |
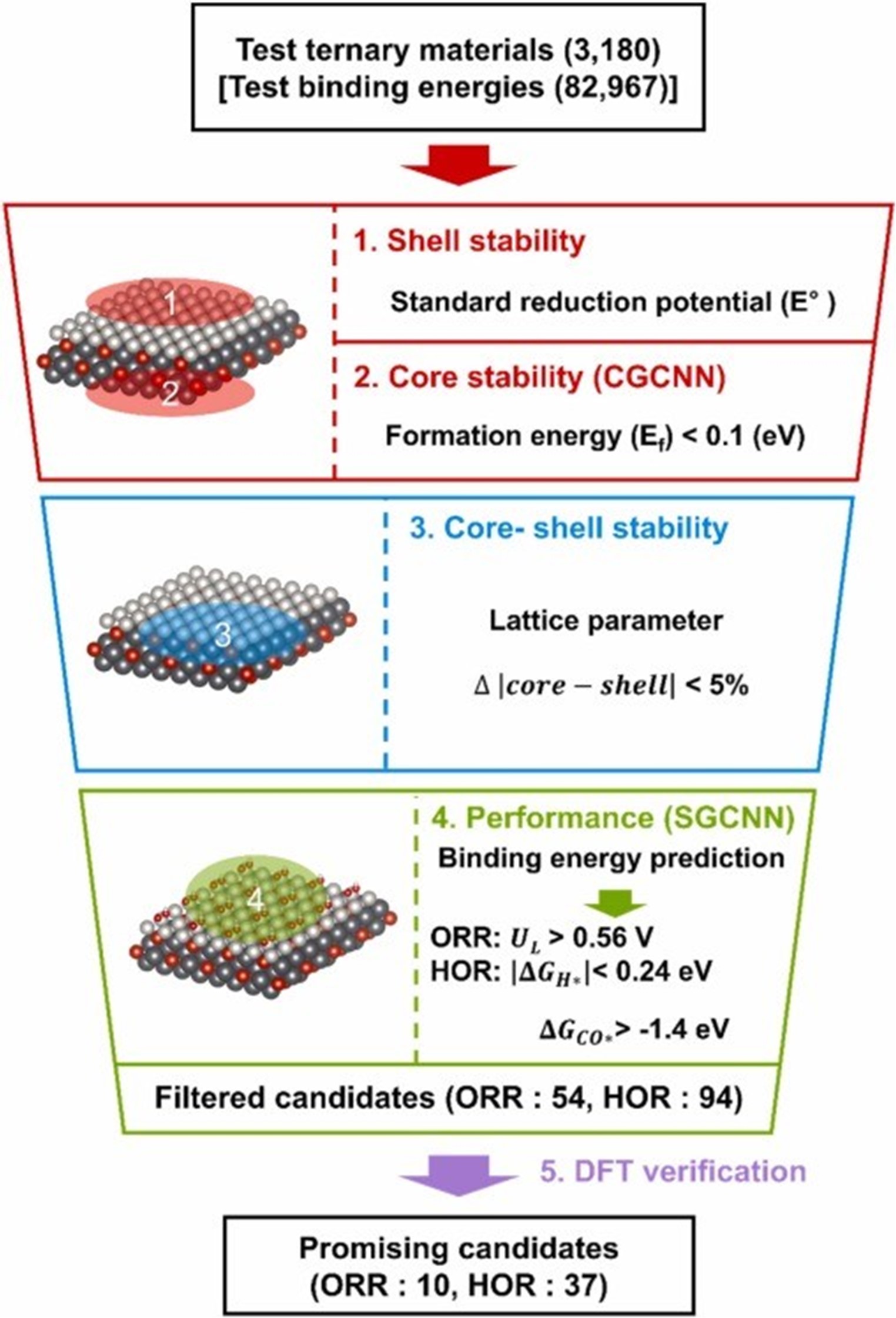 | |
| Machine learning-driven material screening workflow for each anode and cathode of fuel cells. (Image: KIST) | |
| The researchers developed a novel ternary (Cu-Au-Pt) alloy catalyst through experimental validation of 10 catalysts with the potential to outperform platinum catalysts out of approximately 3,200 candidate materials. The catalyst uses only 37% of the element platinum compared to pure platinum catalysts, but the kinetic current density is more than twice as high as that of pure platinum catalysts. The catalyst also exhibits excellent durability, with little degradation after 5,000 stability tests. | |
| "In the future, we plan to continue to build high-quality adsorption energy data and perform more sophisticated AI modeling, which will further improve the success rate of catalytic material development," said Dr. Kim of KIST. The new methodology has the advantage of being immediately applicable not only to catalysts for hydrogen fuel cells, but also to various catalytic reactions such as water electrolysis-based hydrogen production, which is essential for the realization of the hydrogen economy. The team plans to further reduce the unit cost and improve the performance of the developed catalysts through material and system optimization. |
| Source: The Korea Institute of Science and Technology (Note: Content may be edited for style and length) |
